A Review of a Recent Meta-Analysis Study on Obstructive Sleep Apnea
Article information
Abstract
This paper summarizes a recent meta-analysis of various topics in obstructive sleep apnea (OSA). In addition to cardiovascular disease and neurocognitive dysfunction, a wide variety of diseases have been associated with OSA, and associations with cancer have also been reported. Although continuous positive airway pressure is a very effective treatment, the results have shown that it does not reduce the incidence of various complications. It has been reported that uvulopalatopharyngoplasty was effective, and robotic surgery for the tongue root and hypoglossal nerve stimulation were also effective. The effectiveness of various medications to reduce daytime sleepiness has also been demonstrated. Although exercise lowered the apnea-hypopnea index, it was not related to changes in body composition, and it was also reported that exercise combined with weight control were effective. Additionally, interesting and clinically meaningful meta-analysis results were summarized and presented.
INTRODUCTION
Obstructive sleep apnea (OSA) is a disease involving repeated narrowing of the upper respiratory tract during sleep, which causes a decline or discontinuation of respiratory airflow. It is characterized by frequent sleep fragmentation and hypoxia [1]. The incidence rate of OSA is very high, as nearly a billion patients are estimated to exist across the world [2]. Symptoms include severe daytime sleepiness, headache, fatigue, and loss of concentration [3]. It also increases the possibility of hypertension [4], diabetes [5], stroke [6], arrhythmia [7], and other complications as well as car accidents and workplace accidents, which may ultimately result in death. Continuous positive airway pressure (CPAP) is highly effective against OSA. However, due to low compliance, other treatments are often used including surgery and oral appliances.
Meta-analysis is a statistical method of collecting, reviewing, and compiling previously published literature to draw certain conclusions [8]. Meta-analysis is widely used for discussing interesting and controversial topics in the relevant field. As such, the method allows researchers to easily track the key research trends and outcomes in the field, and infer the best results for topics that have not been concluded clearly so far. The purpose of this meta-analysis is to cover various topics related to OSA and provide readers with the latest knowledge and opinions in the field of OSA studies.
LITERATURE SEARCH METHOD
We searched for all meta-analysis studies published on OSA between January 1, 2017 and February 18, 2022, the day when we carried out the search. We searched the studies at PubMed with the search word ‘obstructive sleep apnea,’ using the filter function to narrow down the article types to ‘meta-analysis.’ The search found 384 articles. We reviewed each of them to select interesting studies with high clinical value, and grouped the selected articles across different topics (Fig. 1).
COMPLICATIONS ASSOCIATED WITH OSA
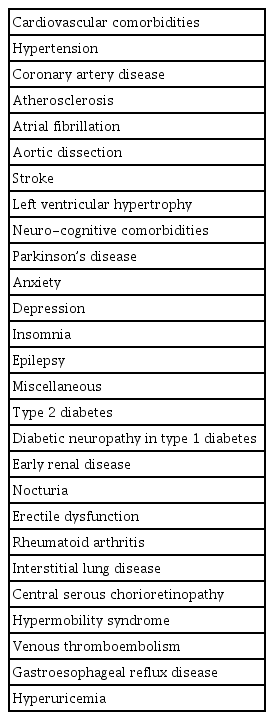
Although exact mechanisms have not been identified, OSA is known to cause or exacerbate various complications. Recent meta-analysis studies have affirmed this association. The most well-known complications include: hypertension, coronary artery diseases and other cardiovascular diseases [6,9-15], the Parkinson’s diseases, anxiety disorders, depression, and other neurological-cognitive disorders [16-20]. Many studies have repeatedly presented consistent findings regarding these complications, and their level of association with OSA is quite high. Studies have also suggested association with lesser-known complications [21-28] including rheumatoid arthritis [29], interstitial lung disease [30], central serous chorioretinopathy [31], and hypermobility syndrome [32] (Table 1). In addition, for some of these complications, metaanalysis articles have reached opposing conclusions about their association with OSA, and researchers continue to report a wide range of other diseases associated with OSA.
OSA AND CANCER
In recent years, researchers have continuously reported associations between OSA and cancer. A meta-analysis on twelve articles (total no. of patients: 862,820) reported that the cancer rate of OSA patients was 0.046% higher than the control group (95% confidence interval [CI]: 0.027–0.065), without clear associations with specific cancers [33]. As for specific cancer types, many studies have reported the association between OSA and melanoma. A meta-analysis study analyzed six of these articles involving 5,276,451 patients, and found that the melanoma risk ratio of OSA patients was 1.71 times higher than the control group (95% CI: 1.08–2.69) [34].
LIMITATIONS OF CPAP
CPAP is one of the most effective treatments for OSA. As such, various studies were conducted on CPAP under the expectation that it will reduce various complications associated with OSA. Of note, as many studies reported that CPAP decreases the blood pressure of OSA patients with hypertension [35], it was logically believed that it can reduce cardiovascular complications as well. A meta-analysis study involving eight randomized controlled trials was designed to test this possibility. This analysis, which involved 5,817 subjects, analyzed the relative risk of cardiovascular diseases in patients treated with CPAP. The overall risk of major cardiovascular diseases was 0.87 (95% CI: 0.70–1.10), and the relative risk of cardiovascular death was 0.94 (95% CI: 0.62–1.43), the relative risk of myocardial infarction was 1.04 (95% CI: 0.79–1.37), and the relative risk of angina was 1.05 (95% CI: 0.51–2.15). CPAP also did not have preventive effect on cardiovascular diseases including heart failure, stroke, and atrial fibrillation [36]. Another study conducted a similar meta-analysis on ten randomized controlled trials (7,266 patients), and the relative risk of acute coronary artery diseases, stroke, and vascular death was 0.77 (95% CI: 0.53–1.13), which suggests that CPAP did not reduce the risk of these diseases comparing with the control group. In sum, in both studies, CPAP did not have preventive effect on various cardiovascular diseases [37]. As both studies only covered randomized controlled trials, the effect of confounding variables during the selection of the CPAP group and the control group seems to be minimal. If we also consider the difference between the duration of CPAP use (4 hours or longer and less than 4 hours) did not produce meaningful differences in either study, it is credible that CPAP does not reduce the occurrence of cardiovascular complications.
Numerous studies have observed the effect of CPAP of type 2 diabetes, which is another main complication associated with OSA. A meta-analysis study involving six randomized controlled trials (581 patients) was performed. A comparison of HbA1c levels at 12 or 24 weeks after CPAP treatment showed mean difference of -0.10 (95% CI: -0.25–0.04), which is not significant. The result is same, even in patients who used CPAP for four hours or longer per day. In addition, the CPAP group and the control group showed similar fasting glucose levels [38].
Excessive abdominal visceral fat is also known as a complication of OSA. In a meta-analysis on five studies that analyzed changes in abdominal visceral fat after CPAP treatment (169 patients), no change in abdominal visceral fat was observed after CPAP treatment (standardized mean difference: 0.00, 95% CI: -0.21–0.21). The result is not different by the CPAP treatment duration, patients’ ages, or their body mass indexes [39].
As discussed above, many studies reported disappointing findings about the preventive effect of CPAP. However, positive findings have been reported as well. CPAP lowers blood pressure [35], and reduces various inflammatory substances [40], cortisol [41], and proteinuria [42]. It also reduces gastroesophageal reflux disease [43], and erectile dysfunction [44].
SURGICAL TREATMENTS OF OSA
There have been many studies that denies the effectiveness of the soft palate surgery as a treatment for OSA. However, there also have been many studies that support the effectiveness of the soft palate surgery [45-48]. A recent meta-analysis study of 59 soft palate surgeries included anterior and lateral palatopharyngoplasty as well as conventional uvulopalatopharyngoplasty, and compared the results before and after the surgeries. The apnea-hypopnea index (AHI) declined from 35.66 to 13.91 after the surgeries (p<0.001), and the Epworth Sleepiness Scale (ESS) also declined from 11.65 to 5.08 (p<0.001) [49].
Many studies reported findings on OSA surgeries using robots, which are mainly used to dissect the root of the tongue. A meta-analysis on 31 of these studies (1,693 patients) found a 24.3 decline in the AHI (95% CI: 21.69–26.81), a 7.92 decline in ESS (95% CI: 6.50–9.34), a 6.04% increase in the lowest oxygen saturation (95% CI: 3.05–9.03), and 69% in a surgery success rate (95% CI: 64–79). However, these findings provide low evidence for proving the effectiveness of the robot surgeries, because most of the robot surgeries were performed along with soft palate surgeries and the studies did not include any control group [50].
Recently one of the most notable surgical treatments for OSA is the hypoglossal nerve stimulation therapy. This therapy inserts an electrode in the human body to stimulate the hypoglossal nerves to resolve tongue root blocking. It has been recognized as effective in numerous studies and was approved by the United States Food and Drug Administration in 2014. Korea is also hurrying the approval process. A meta-analysis was conducted on 15 studies (808 patients) published on this topic, which found a 24.9 decline in the AHI (95% CI: 21.1– 28.5) [51].
Before the hypoglossal nerve stimulation therapy was developed, transcutaneous electrical stimulation has been attempted in some cases to stimulate the hypoglossal nerves. Conflicting findings have been reported regarding this method. In a meta-analysis on five studies (87 patients) on this method, the AHI decline by 16.5 (95% CI: 7.7–25.1) [51].
MEDICATIONS FOR OSA
In the past, modafinil was widely used for treating narcolepsy and hypersomnia associated with OSA, and a large number of studies were performed on the topic [52,53]. Since then, various medications have been developed for OSA. Solriamfetol is a norepinephrine–dopamine reuptake inhibitor developed by a Korean pharmaceutical company [54,55]. Five randomized controlled trials (1,777 patients) were conducted regarding esolriamfetol, and a meta-analysis on these studies found that solriamfetol lowered the ESS by an average of 3.74 points (95% CI: 3.09–4.38) [56]. Pitolisant (histamine 3 receptor antagonist/inverse agonist) is a new medication recently developed for OSA. Four randomized controlled trials (678 patients) were conducted regarding pitolisant, and a metaanalysis on these studies found a 2.86 decline in the ESS (95% CI: 1.96–3.75) and a 3.14-minutes increase in sleep latency (95% CI: 2.18–4.11) [57].
Non-benzodiazepine hypnotics, such as zolpidem, zaleplon, and eszopiclone, improve sleep onset and sleep continuity without changing the overall sleep stages [58,59]. As such, these drugs were expected to increase CPAP compliance of OSA patients. According to a meta-analysis on this issue, nonbenzodiazepine hypnotics increased the average time for CPAP use by 0.62 hour (95% CI: 0.26–0.98) and increased the days of CPAP use by 12.08% (95% CI: 5.27–18.88). Among them, eszopiclone proved to be the most effective [60].
Some studies found that serotonin leads to the contraction of the genioglossus and the upper airway dilating muscles by stimulating the hypoglossal nerves [61,62]. A meta-analysis on these studies concluded that serotonin is not recommended as an OSA treatment. Although some studies reported a decline in the AHI comparing with the control group, they did not have a sufficient sample size and have a high risk of bias. Also, serotonin itself is highly likely to cause adverse side effects [63].
EFFECT OF EXERCISE IN OSA TREATMENT
Exercise is emphasized as a key element in the treatment of OSA patients. It is widely known that exercise improves OSA by reducing weight of patients. Some studies have shown that changes in body composition plays a certain part as well; exercise lowers the patient’s body fat and increases the muscle mass, reducing the obstruction of the upper respiratory tract and finally reducing OSA. A meta-analysis on this topic found that OSA patients with exercise have a significant reduction in the body body mass index (-0.55 kg/m2, 95% CI: -0.95 to -0.15) and a significant decline in the AHI (-8.06/hour, 95% CI: -10.47 to -5.66). However, no significant change was observed in body fat. To summarize, while exercise lowers the AHI, it is not an effect from changes in body composition [64].
According to a meta-analysis on the effect of weight loss and lifestyle changes in OSA patients, the effect of diet control, exercise, sleep environment, and cessation of alcohol consumption and smoking had different effects depending on the specific method and the targets. However, weight loss by diet control and exercise proved most effective for adult males with severe or moderate OSA [65].
OTHER TREATMENTS
Some studies concerned that, in temporomandibular disorder patients with OSA, the mandibular advancement device may exacerbate their temporomandibular disorder symptoms. However, a meta-analysis on the topic found that use of the device did not severely exacerbate the patients’ symptoms [66].
Various devices have been developed that change the sleeping positions of patients with severe OSA in the supine position, and numerous studies have been performed on the devices. A meta-analysis found that some of these devices were partially effective, but not as effective as CPAP [67].
Nasal congestion has significant effect on compliance with CPAP. It has been expected that the nasal steroid spray may improve CPAP compliance or increase the time of its use by reducing nasal congestion in OSA patients. A meta-analysis on this issue concluded that nasal steroid spray increased the time of a single CPAP use by 0.4 hour on average, not statistically significant due to the small sample size [68].
OTHER FINDINGS
Alcohol, caffeine, and tobacco have been suspected of exacerbating OSA symptoms. According to a meta-analysis on this issue, alcohol consumption increases OSA occurrence by 1.33 times (95% CI: 1.10–1.62), whereas smoking was not associated with OSA. There was no sufficient data to verify the association between caffeine and OSA [69].
A meta-analysis was performed with 21 studies (54,276 patients) on the effect of OSA on the prognosis of COVID-19 patients. OSA proved to be a seriously negative prognostic factor, as it increased the progress of severe COVID-19 by 1.70 times (95% CI: 1.18–2.45), admission to intensive care units (ICUs) by 1.76 times (95% CI: 1.51–2.05), use of ventilators by 1.67 times (95% CI: 1.48–1.88), and mortality by 1.74 times (95% CI: 1.39–2.19) [70].
Some studies demonstrated that OSA patient have olfactory dysfunction. A meta-analysis on these studies found that threshold, discrimination, identification (TDI) score of OSA patients (24.3±5.6) was lower than that of the control group (30.7±6.0). The AHI was inversely correlated with the TDI score. However, the correlation was minimal [71].
It has been also reported that OSA patients are more likely to be involved in workplace accidents due to daytime sleepiness. A meta-analysis on this issue reported a 1.64 time increase in workplace accidents in OSA patients comparing with the control group (95% CI: 1.24–2.16), and a 1.68 times increase in accidents involving professional drivers with OSA (95% CI: 1.14–2.49) [72].
The telomere is a region of repetitive nucleotide sequences at the end of the chromosome. Telomere shortens each time a cell divides that is associated with the aging of the cells. According to a meta-analysis study on the relationship between OSA and the length of the telomere, the telomere of OSA patients was shorter than that of the control group, and the gap increased in older patients. As such, the authors emphasized the importance of early treatment for OSA [73].
A meta-analysis on the association between OSA and pregnancy-related diseases found that the OSA increases gestational hypertension by 1.97 times, gestational diabetes by 1.55 times, pre-eclampsia by 2.35 times, the frequency of Cesarean section operation by 1.42 times, post-operation wound infection by 1.87 times, pulmonary edema by 6.35 times, preterm birth by 1.62 times, and ICU admission of newborns by 1.28 times [74].
CONCLUSIONS
OSA auses various diseases including cancer and the previously known complications such as cardiovascular diseases and neurological-cognitive disorders. While CPAP is widely known as the most effective treatment, studies have demonstrated its limitations in preventing cardiovascular diseases, diabetes, and other complications. The soft palate surgery has clear benefits, and hypoglossal nerve stimulation, various medications, and exercise improve the effects of OSA treatment.
Notes
Ethics Statement
Ethical approval and informed consents does not apply to this article.
Availability of Data and Material
All data generated or analyzed during the study are included in this published article.
Conflicts of Interest
The authors have no potential conflicts of interest to disclose.
Author Contributions
Conceptualization: Jae Hoon Cho. Data curation: Jae Hoon Cho. Formal analysis: Jae Hoon Cho. Funding acquisition: Jae Hoon Cho. Investigation: all authors. Methodology: all authors. Project administration: Jae Hoon Cho. Resources: Jin Kook Kim, Jae Hoon Cho. Software: Jae Hoon Cho. Supervision: Jae Hoon Cho. Validation: all authors. Visualization: Jae Hoon Cho. Writing—original draft: Bo Yoon Choi, Jae Hoon Cho. Writing—review & editing: all authors.
Funding Statement
None